The collision theory
The Collision Theory (not to be confused with the Particle Theory or the Kinetic Theory of Matter, see at the end for more details or in the Year 8 Chemistry Weebly) is a simple and useful model that explains how chemical reactions occur and why reaction rates differ for different reactions. It was proposed around 1917.
The Collision Theory states that:
In the next section, the effect of a number of factors on rates of reaction is explained in terms of the collision theory statements.
The Collision Theory states that:
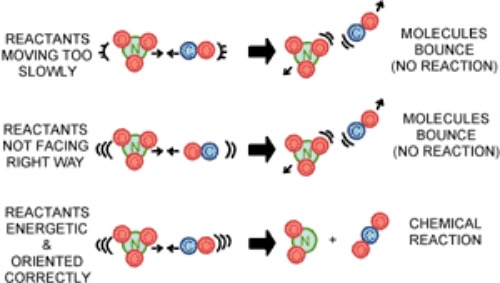
- For a chemical reaction to occur, reactants must collide. The more often reactant molecules collide, the more often they react with one another, and the faster the reaction rate.
- If reactant particles do not collide with enough energy and with right orientation, they will not react together. In reality, only a small fraction of the overall collisions, are effective collisions, that result in a chemical reaction.
- This is because only a small number of molecules have the right orientation and the right amount of energy at the moment of impact to break the existing bonds and form new ones.
- The energy barrier reactant particles must overcome to break bonds is called the activation energy (Ea).
In the next section, the effect of a number of factors on rates of reaction is explained in terms of the collision theory statements.
Watch the videoclip below to consolidate your understanding of the collision theory.
What is activation energy?
There is an easy way to think about the meaning of activation energy: have you ever tried to bake cupcakes in an oven at 80oC? No matter how long you leave the cupcakes in the oven, they will not bake and turn up in the soft and spongy treat you expect. There is simply not enough heat energy available to the batter for effective collisions to happen between the ingredients. At best, you will only end up with a dried up batter.
Another analogy is that of a mountain. If you want to go over a mountain through a steep road, your vehicle should have sufficient power, regardless of how easy it will be to go down on the other side of the mountain.
Finally, you know that hydrogen reacts violently with oxygen to produce water and a lot of heat. However, no reaction will occur in a mixture of hydrogen and oxygen until you provide the initial energy the molecules of gas need to react together.
The video below summarises the main ideas about effective collisions and activation energy.
There is an easy way to think about the meaning of activation energy: have you ever tried to bake cupcakes in an oven at 80oC? No matter how long you leave the cupcakes in the oven, they will not bake and turn up in the soft and spongy treat you expect. There is simply not enough heat energy available to the batter for effective collisions to happen between the ingredients. At best, you will only end up with a dried up batter.
Another analogy is that of a mountain. If you want to go over a mountain through a steep road, your vehicle should have sufficient power, regardless of how easy it will be to go down on the other side of the mountain.
Finally, you know that hydrogen reacts violently with oxygen to produce water and a lot of heat. However, no reaction will occur in a mixture of hydrogen and oxygen until you provide the initial energy the molecules of gas need to react together.
The video below summarises the main ideas about effective collisions and activation energy.
Disambiguation: how are the Particle Theory and Collision Theory different?
The Particle Theory and the Collision Theory are sometimes confused. While the Collision Theory explains how the speed of chemical reaction can be acted upon, the Particle Theory is useful to understand the physical properties of the various states of matter (solid, liquid, gas).
The Particle Theory (a.k.a. Kinetic Theory of Matter) states that all matter consists of many, very small particles which are constantly moving or in a continual state of motion. The degree to which the particles move is determined by the amount of energy they have and their relationship to other particles. The particles might be atoms, molecules or ions. Use of the general term 'particle' means the precise nature of the particles does not have to be specified.
Particle Theory helps to explain properties and behaviour of materials by providing a model which enables us to visualise what is happening on a very small scale inside those materials. For example, it helps to understand the structure of the different states of matter and what happens during physical changes such as melting, boiling and evaporating.
The Particle Theory and the Collision Theory are sometimes confused. While the Collision Theory explains how the speed of chemical reaction can be acted upon, the Particle Theory is useful to understand the physical properties of the various states of matter (solid, liquid, gas).
The Particle Theory (a.k.a. Kinetic Theory of Matter) states that all matter consists of many, very small particles which are constantly moving or in a continual state of motion. The degree to which the particles move is determined by the amount of energy they have and their relationship to other particles. The particles might be atoms, molecules or ions. Use of the general term 'particle' means the precise nature of the particles does not have to be specified.
Particle Theory helps to explain properties and behaviour of materials by providing a model which enables us to visualise what is happening on a very small scale inside those materials. For example, it helps to understand the structure of the different states of matter and what happens during physical changes such as melting, boiling and evaporating.