atomic structure and subatomic particles
|
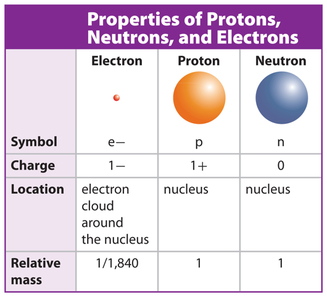
Some of the important properties of the three subatomic particles making up atoms are summarised in the right hand side table.
However protons and neutrons are not elementary particles. More details on their inner composition can be found on the Chemistry 9 Weebly, in the Important Experiments section. |
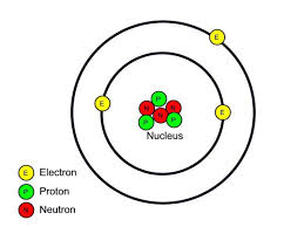
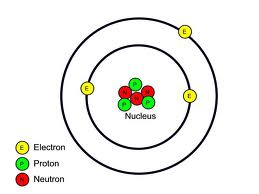
Anatomy of an atom
Mass of particles
Electrical charge of particles
Size of atoms
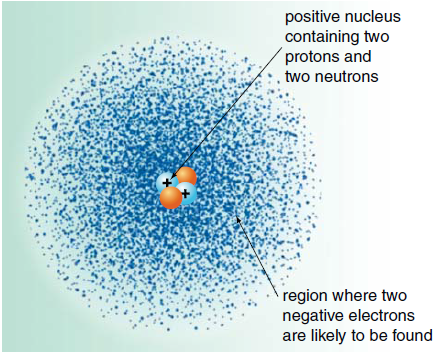
- An atom consists of a positively charged nucleus orbited by fast moving negatively charged particles called electrons.
- The nucleus contains only two types of particles: protons and neutrons. These are collectively called nucleons.
- The number of protons in an atom defines its atomic number and from there its position in the periodic table and the name of the corresponding element.
- The number of nucleons is called the mass number of the atom. It is similar but not the same as the relative atomic mass of an element which is most often a decimal number.
- Electrons orbit the nucleus at a very large distance compared to the size of the nucleus, and at very high speed.
Mass of particles
- Protons and neutrons have a mass of approximately 1 atomic mass unit or 1 u (see Chemistry 9 Weebly for the definition of atomic mass unit).
- Compared to the mass of the protons and neutrons, the electrons have negligible mass (only 1/1840th u) and thus can be ignored when carrying out calculations involving mass.
Electrical charge of particles
- Protons carry a single positive electrical charge (tip: the letter 'p' in proton also stands for positive) and the electrons carry a single negative electrical charge. Neutrons carry no charge, hence their name.
- Although protons should repel each other due their electrical charge, and make the nucleus unstable, they are held together by a force called the nuclear force. Neutrons and protons are affected by the nuclear force almost identically. Moreover, neutrons act as "spacers" between protons, preventing protons from being too close from each other, therefore reducing the force of repulsion.
- Overall, atoms are neutral, which means that they must have as many positive charges as negative charges, or in other words as many electrons as protons.
Size of atoms
- Individual atoms can be visualised as spheres. The size of an atom is usually associated to its radius. The unit used to describe the very small length values involved at the atomic scale is the nanometer, abbreviated as nm. 1 nm = 0.000 000 001 m, or a billionth of a meter.
- Atomic radius vary between about 0.1 nm (for hydrogen atoms) to about 0.4 nm. The atomic radius of the elements in the Periodic Table lie between these 2 values.
Representation of an atom: the electron cloud model
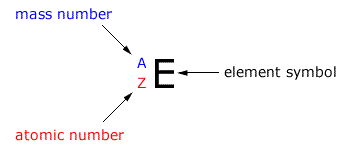
AZE nomenclature
It is a system of abbreviating the atoms to show the fundamental sub-atomic particles within.
|
Example: How many protons and electrons does an atom of iron contain?
Answer: The atomic number (A) of iron is 26 therefore it contains 26 protons.
The number of electrons = number of protons, therefore there are 26 electrons.
Answer: The atomic number (A) of iron is 26 therefore it contains 26 protons.
The number of electrons = number of protons, therefore there are 26 electrons.
Did you know?
In nuclear physics language, nucleons belong to a larger group of particles called hadrons. The name hadron is found in the full name of the world's larger particle accelerator located in Switzerland: The Large Hadron Collider or LHC. Click here if you would like to read, hear and learn more about sub-atomic particles, particle accelerators and the LHC.