Chemical reactions
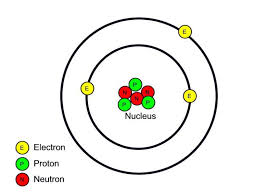
- The outer most shell is called valence shell and the electrons in the valence shell are called valence electrons. The reactivity of elements is determined by the number of valence electrons.
- With the notable exception of elements belonging to the 18th group which have a complete valence shell (noble gases), all elements in their atomic state will almost always react in a way to complete their valence shell, either by gaining or losing electrons. By completing valence shells, atoms become ions and achieve extra stability compared to the elemental form.
- Metal elements always lose electrons to become cations. Non-metal elements most often gain electrons to become anions. Semi-metals