chemical and physical properties trends
In a nutshell
- A trend indicates a general direction in which a physical or chemical property is developing or changing.
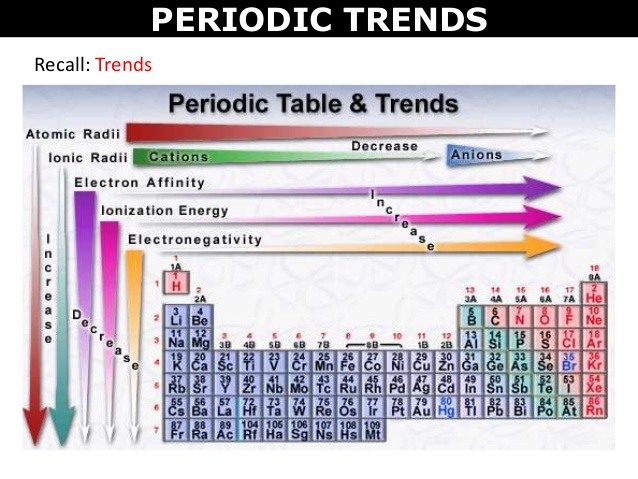
- The image below summarises trends in the Periodic Table for atomic radii, ionic radii and first ionisation energy (electron affinity and electronegativity are more advanced concepts that will be explored in Year 11 Chemistry). Remember that a trend does not account for possible exceptions.
Atomic radii
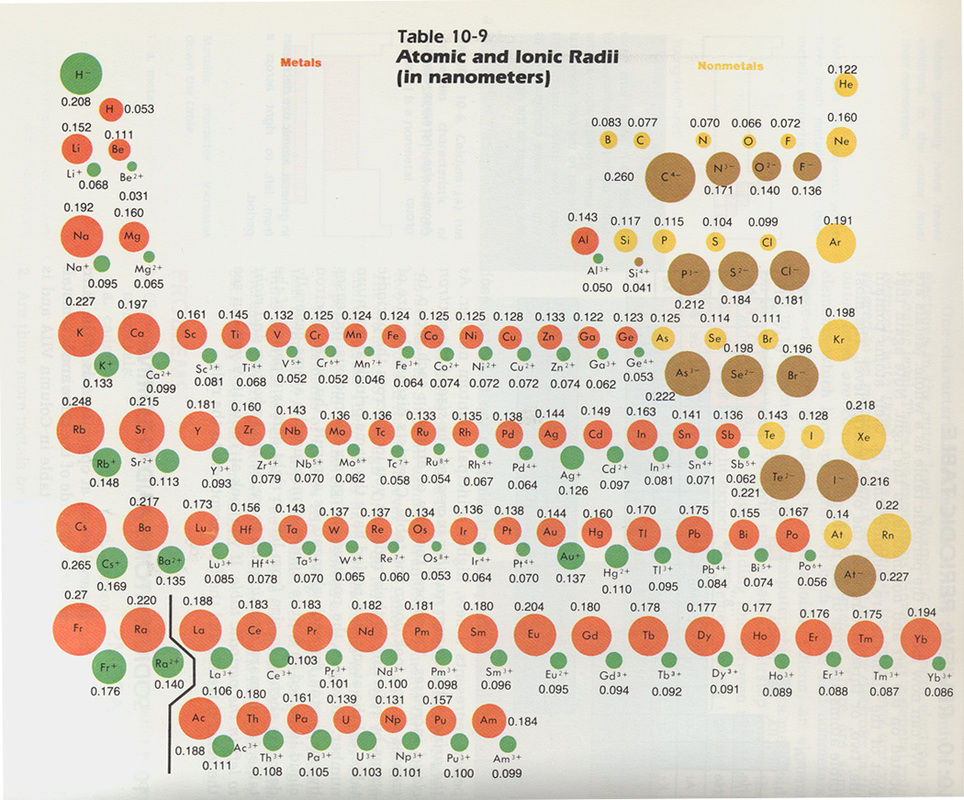
The size of atoms is usually best described by their radius. With the exception of hydrogen (with an atomic radius of 0.11 nanometres or nm), helium (the most top-right element) has the smallest radius (0.14 nm) while francium has the largest value (0.35 nm). All other elements have an atomic radius lying between these 2 values.
General trend: atomic radius of elements decreases from left to right within a period, and increases from top to bottom. In other words, the atomic radius increases from the top right of the Periodic Table to the bottom left.
Note: 1 namometre or 1 nm represents a billionth of a metre.
Ionic radii vs atomic radii
Ions are atoms with a different number of protons and electrons. A difference in the number of protons and electrons is usually obtained by adding or removing electrons from the most outer electron shell, known as the valence shell.
When an atom loses one or more electrons, it becomes positively charged and is known as cation. Conversely, when an atom gains one or more electrons, it becomes negatively charged and is called anion.
General trend: Cations have always a smaller atomic radius than the neutral atom they are made from while anions have always a larger atomic radius than the atom they originate from.
The size of atoms is usually best described by their radius. With the exception of hydrogen (with an atomic radius of 0.11 nanometres or nm), helium (the most top-right element) has the smallest radius (0.14 nm) while francium has the largest value (0.35 nm). All other elements have an atomic radius lying between these 2 values.
General trend: atomic radius of elements decreases from left to right within a period, and increases from top to bottom. In other words, the atomic radius increases from the top right of the Periodic Table to the bottom left.
Note: 1 namometre or 1 nm represents a billionth of a metre.
Ionic radii vs atomic radii
Ions are atoms with a different number of protons and electrons. A difference in the number of protons and electrons is usually obtained by adding or removing electrons from the most outer electron shell, known as the valence shell.
When an atom loses one or more electrons, it becomes positively charged and is known as cation. Conversely, when an atom gains one or more electrons, it becomes negatively charged and is called anion.
General trend: Cations have always a smaller atomic radius than the neutral atom they are made from while anions have always a larger atomic radius than the atom they originate from.
First ionisation energy
Removing an electron from a neutral atom costs energy. The minimum energy required to remove the first electron from an atom is called first ionisation energy.
General trend: the first ionisation energy increases from left to right within a period, and decreases from top to bottom in a group. In other words, the first ionisation energy increases from the bottom left to the top right of the Periodic Table, that is in opposite direction to the atomic radius.
Melting and boiling points
Although elements occur in different states at room temperature, they almost all have a melting point and boiling point, the exception being carbon that sublimes (changes directly from the solid state to gaseous state).
There is no obvious trend for melting and boiling but it can be roughly found that they increase then decrease from left to right, reaching a maximum value for elements of group 14 for periods 2 &3, and for elements of group 6 from period 4. No clear trend is found from top to bottom, although trends exist within individual groups.
Removing an electron from a neutral atom costs energy. The minimum energy required to remove the first electron from an atom is called first ionisation energy.
General trend: the first ionisation energy increases from left to right within a period, and decreases from top to bottom in a group. In other words, the first ionisation energy increases from the bottom left to the top right of the Periodic Table, that is in opposite direction to the atomic radius.
Melting and boiling points
Although elements occur in different states at room temperature, they almost all have a melting point and boiling point, the exception being carbon that sublimes (changes directly from the solid state to gaseous state).
There is no obvious trend for melting and boiling but it can be roughly found that they increase then decrease from left to right, reaching a maximum value for elements of group 14 for periods 2 &3, and for elements of group 6 from period 4. No clear trend is found from top to bottom, although trends exist within individual groups.
Click here to access an interactive Periodic Table that gives the accepted values for density, atomic size, melting and boiling points and first ionisation energy of elements.