States of matter
Physical characteristics
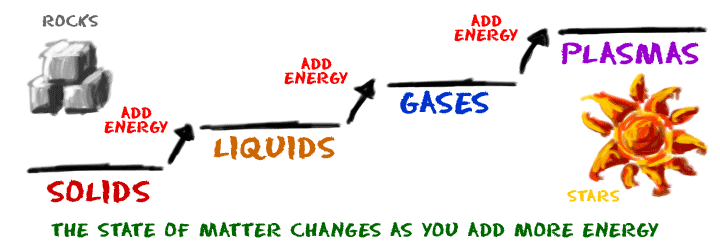
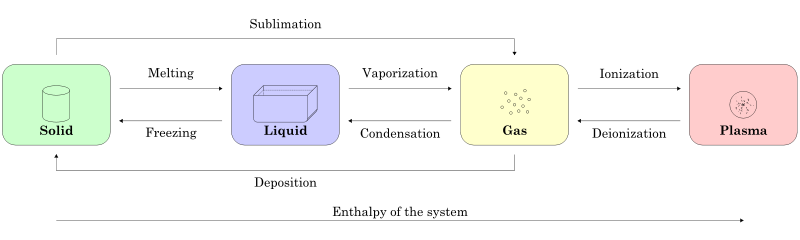
A state of matter is one of the distinct forms that matter takes on. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma.
Note: The term phase is sometimes used as a synonym for state of matter, but a system can contain several immiscible phases of the same state of matter
- Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place.
- Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely.
- Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place.
- Matter in the plasma state has variable volume and shape, but as well as neutral atoms, it contains a significant number of ions and electrons, both of which can move around freely. Plasma is the most common form of visible matter in the universe.
Note: The term phase is sometimes used as a synonym for state of matter, but a system can contain several immiscible phases of the same state of matter
Particle theory
The particle theory (aka kinetic theory) helps explain the properties of matter, for example what happens during physical changes such as melting, boiling and evaporating.
This theory states that all matter consists of many, very small particles which are constantly moving or in a continual state of motion. The degree to which the particles move is determined by the amount of energy they have and their relationship to other particles.
The particles might be atoms, molecules or ions. We will use the general term 'particle' means the precise nature of the particles does not have to be specified.
Particle theory provides a model which enables us to visualise what is happening on a very small scale inside those materials. As a model it is useful because it appears to explain many phenomena but as with all models it does have limitations.
This theory states that all matter consists of many, very small particles which are constantly moving or in a continual state of motion. The degree to which the particles move is determined by the amount of energy they have and their relationship to other particles.
The particles might be atoms, molecules or ions. We will use the general term 'particle' means the precise nature of the particles does not have to be specified.
Particle theory provides a model which enables us to visualise what is happening on a very small scale inside those materials. As a model it is useful because it appears to explain many phenomena but as with all models it does have limitations.