electron configuration
In a nutshell:
- Electrons are arranged around the nucleus in shells of well defined energy. The number of electrons of neutral atoms corresponds to the atomic number.
- Each shell can only contain a given number of electrons, that corresponds to the number of elements in the corresponding period. Hence, the maximum number of electrons per shell, in order of increasing shell number (from 1 to 4) is 2,8,18,32.
- Electron shells consist of sub-shells denoted s, p, d and f.
- When adding electrons around a nucleus, the electrons are always placed in the shell with the lowest number first, then the second lowest one, and so on.
- Within a shell, sub-shells are generally filled in the order s, p, d and f.
Electron shell (energy level)
|
You learnt in Year 9 that electrons are arranged around the nucleus in energy levels, or electron shells.The maximum number of electrons per shell, in order of increasing shell number (from 1 to 4) was said to be respectively 2, 8, 8, and 18. An atom will be made of the same number of electron shells as the number of period where it is found in the Periodic Table. The video above is meant to refresh your memory about the electron configuration notation seen in Year 9. However, this representation is only partly true. To understand why, the concept of subshell or sublevel must be introduced.
|
|
Electron subshells (or sublevels)
Each electron shell consists of subshells. The first shell or energy level is made up of only one subshell, the second shell consists of two subshells, the third shell has three subshells, and so on for higher energy levels. The relationship is that there are as many sublevels per level as its number.
Currently, there are four types of subshells known to be used by electrons in atoms up to element 118, the heaviest atom ever produced. Sublevels differ by the shape of the maximum number of electrons they can hold. To differentiate between them, each type of subshell is denoted by a letter.
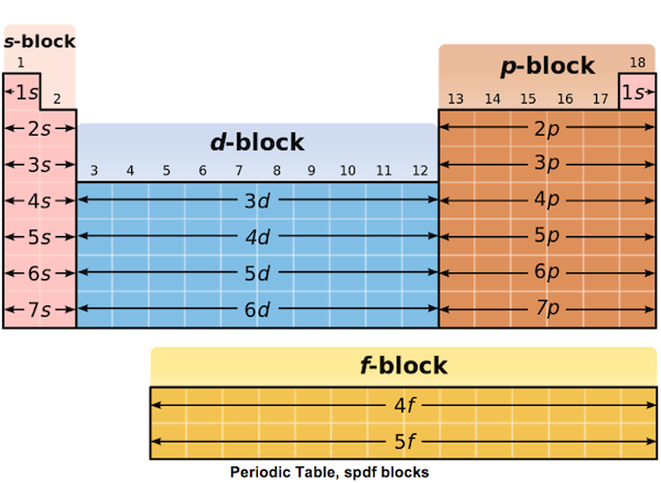
This more detailed way of describing the arrangement of electrons in atoms is reflected in a representation of the Periodic Table (below) that shows the elements grouped in blocks: the s-block, the p-block, the d-block and the f-block.
Currently, there are four types of subshells known to be used by electrons in atoms up to element 118, the heaviest atom ever produced. Sublevels differ by the shape of the maximum number of electrons they can hold. To differentiate between them, each type of subshell is denoted by a letter.
- any s subshell can hold a maximum of 2 electrons. All energy levels have an s sub-shell.
- any p subshell can hold up to 6 electrons. All energy levels except the first one have a p sub-shell.
- any d subshell can hold up to 10 electrons. Energy levels higher than two have a d sub-shell.
- any f subshell can hold up to 14 electrons. This type of sublevel is found from the fourth energy level.
This more detailed way of describing the arrangement of electrons in atoms is reflected in a representation of the Periodic Table (below) that shows the elements grouped in blocks: the s-block, the p-block, the d-block and the f-block.
As can be seen above, each subshell has its own name.
The block to which an element belongs indicates that its valence electrons are found in the corresponding subshell. For example, since phosphorus, group 15, belongs to the p-block, its valence electrons are found in the p subshell. Knowing what sub-shell the valence electrons are in has strong implications on the reactivity of an element.
- The s subshell of the first energy level is called 1s. Any atom contain at least the subshell 1s, starting with hydrogen atoms up to oganesson atoms (atoms of element 118).
- The s and p subshells of the second shell, or second energy level, are denoted 2s and 2p. Any element from lithium has electrons in the 2s subshells and any element from boron has electrons in the 2p subshells.
- The third shell has three subshells: 3s and 3p and 3d.
The block to which an element belongs indicates that its valence electrons are found in the corresponding subshell. For example, since phosphorus, group 15, belongs to the p-block, its valence electrons are found in the p subshell. Knowing what sub-shell the valence electrons are in has strong implications on the reactivity of an element.
Electron configuration using the subshell notation
The modern way of writing electron configurations uses a combination of the name of these subshells and the number of the electron shell.
Consider the 2 elements of the first period of the Periodic Table, hydrogen and helium. They have respectively 1 and 2 electrons in the first and only sub-shell (s) of the first energy level. To indicate this, the following notation is used to indicate their electron configuration: 1s1 for hydrogen and 1s2 for helium, which used to be written 1 and 2 in the former notation learnt in Year 9. Lithium atoms, with 3 electrons, have 2 electrons in the first sub-shell (s) of the first energy level and 1 electron in the first sub-shell (s) of the second energy level. The electron configuration of lithium atoms is written 1s2 2s1 (or 2,1 in the simpler notation).
Boron atoms, with 5 electrons, have the electron configuration 1s2 2s2 2p1, which means 2 electrons in the first sub-shell of the first energy level, 2 electrons in the first sub-shell of the second energy level and the fifth electron in the second sub-shell of the second energy level. The simpler notation used to be 2,3.
Because the subshell 3d is higher in energy than the subshell 4s, the subshell 4s is going to be filled before the subshell 3d. This is why the third energy level is often said to accept up to 8 electrons, although it can actually contain up to 18 electrons and this is the reason why the third period of the periodic table contains only 8 elements. Argon, the last element of the third has the electron configuration 1s2 2s2 2p6 3s2 3p6. The first element of the fourth period, potassium, which has one extra electron compared to argon, has the configuration 1s2 2s2 2p6 3s2 3p6 4s1.
(Read the EXtra bits for EXperts section at the bottom of this page for more details on the Electron Cloud Model.)
Consider the 2 elements of the first period of the Periodic Table, hydrogen and helium. They have respectively 1 and 2 electrons in the first and only sub-shell (s) of the first energy level. To indicate this, the following notation is used to indicate their electron configuration: 1s1 for hydrogen and 1s2 for helium, which used to be written 1 and 2 in the former notation learnt in Year 9. Lithium atoms, with 3 electrons, have 2 electrons in the first sub-shell (s) of the first energy level and 1 electron in the first sub-shell (s) of the second energy level. The electron configuration of lithium atoms is written 1s2 2s1 (or 2,1 in the simpler notation).
Boron atoms, with 5 electrons, have the electron configuration 1s2 2s2 2p1, which means 2 electrons in the first sub-shell of the first energy level, 2 electrons in the first sub-shell of the second energy level and the fifth electron in the second sub-shell of the second energy level. The simpler notation used to be 2,3.
Because the subshell 3d is higher in energy than the subshell 4s, the subshell 4s is going to be filled before the subshell 3d. This is why the third energy level is often said to accept up to 8 electrons, although it can actually contain up to 18 electrons and this is the reason why the third period of the periodic table contains only 8 elements. Argon, the last element of the third has the electron configuration 1s2 2s2 2p6 3s2 3p6. The first element of the fourth period, potassium, which has one extra electron compared to argon, has the configuration 1s2 2s2 2p6 3s2 3p6 4s1.
(Read the EXtra bits for EXperts section at the bottom of this page for more details on the Electron Cloud Model.)
The first video clip below explains how many sublevels (or subshells) are found in each energy level and how these sublevels are arranged in order of increasing energy. The second one details how to write the electrons condifuration of elements using the subshell notation
|
Check you understanding:
1) What is the difference between energy level and electron shell? 2) Name the 4 different types of subshells. 3) How many subshells are making up the fourth energy level? 4) How many electrons can a p subshell contain? 5) Where is the s-block in the Periodic Table? 6) What element has one electron in the 3d sublevel? 7) Why is the d sub-shell of the third energy level filled after the s subshell of the fourth energy level? 8) Give the electron configuration of atoms of the elements sulfur and titanium using the subshell notation. |
The video clip below explains how to decide the relative energy of electron subshells using the Periodic Table.
Valence electrons
The electrons in the outermost occupied shell determine the chemical properties of the atom and are called valence electrons. The outermost shell itself is called the valence shell of an atom. In this way, the chemical reactivity of an element is highly dependent upon its electronic configuration, particularly the number of electrons in the valence shell.
Remember the following trend: Elements belonging to the same group have the same number of valence electrons. It enables you to decide the number of valence electrons for all elements belonging to the s-block and p-block of the Periodic Table. For instance, barium atoms have 2 valence electrons since it belongs the same group as beryllium and we know that beryllium atoms have 2 valence electrons.
The number of valence electrons for elements belonging to the d-block is not as straightforward and is not be addressed here.
Remember the following trend: Elements belonging to the same group have the same number of valence electrons. It enables you to decide the number of valence electrons for all elements belonging to the s-block and p-block of the Periodic Table. For instance, barium atoms have 2 valence electrons since it belongs the same group as beryllium and we know that beryllium atoms have 2 valence electrons.
The number of valence electrons for elements belonging to the d-block is not as straightforward and is not be addressed here.
Activity: Determine the number of valence electrons in atoms of the following elements: helium
oxygen
magnesium
indium
tellurium
bismuth
cesium
bromine
oganesson
tenessine
moscovium
oxygen
magnesium
indium
tellurium
bismuth
cesium
bromine
oganesson
tenessine
moscovium
EXtra bits for EXperts: The Electron Cloud Model
In 1926, Schrodinger proposed that the exact path of an electron cannot be known accurately at any instant, unlike in the case of planets around a star. He devised mathematical equations describing the likelihood of finding an electron in a certain position around the nucleus. The Electron Cloud Model (aka quantum mechanical model, or the Schrodinger equation) predicts the likely position of the location of the electron based on a function of probabilities. The probability function basically describes a cloud-like region of space where the electron is likely to be found, hence the name. As the distance from the nucleus increases, the probability to find electrons decreases, but, interestingly, without ever becoming 0.
The region of space where the probability to find the electrons is larger than 90 % is known as atomic orbital, since it is the most likely location where an orbiting electron will be found. Any orbital acan only contain up to 2 electrons.
Orbitals are grouped in subshells, except for subshells s that have only one orbital. Subshells p consist of 3 orbitals and subshells d have 5 orbitals. Since the probability to find electrons decreases as the distance from the nucleus increases, atomic orbitals are centered on the nucleus.
In 1926, Schrodinger proposed that the exact path of an electron cannot be known accurately at any instant, unlike in the case of planets around a star. He devised mathematical equations describing the likelihood of finding an electron in a certain position around the nucleus. The Electron Cloud Model (aka quantum mechanical model, or the Schrodinger equation) predicts the likely position of the location of the electron based on a function of probabilities. The probability function basically describes a cloud-like region of space where the electron is likely to be found, hence the name. As the distance from the nucleus increases, the probability to find electrons decreases, but, interestingly, without ever becoming 0.
The region of space where the probability to find the electrons is larger than 90 % is known as atomic orbital, since it is the most likely location where an orbiting electron will be found. Any orbital acan only contain up to 2 electrons.
Orbitals are grouped in subshells, except for subshells s that have only one orbital. Subshells p consist of 3 orbitals and subshells d have 5 orbitals. Since the probability to find electrons decreases as the distance from the nucleus increases, atomic orbitals are centered on the nucleus.
|
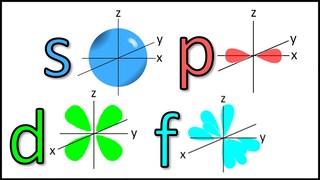
The diagram on the right shows the typical shapes of atomic orbitals. While atomic orbitals s have a spherical ball shape, other have unexpected shapes such as dumbbell shape for p orbitals or a flower shape for d orbitals. You will learn more about this in Year 11. |
Generic shape of the main types of atomic orbitals
|