Covalent bonding
Covalent bonding happens when non-metallic atoms bond with each other. Non-metals have the ability to remove electrons from metals but they can’t do this to other nonmetals. Instead, they share some of their outer-shell electrons.
Covalent bonds happen when two non-metals share one or more pairs of outer-shell electrons. If one pair is shared, then one electron from each atom forms the bond. The shared grip on these electrons holds the two atoms together.
Covalent bonds happen when two non-metals share one or more pairs of outer-shell electrons. If one pair is shared, then one electron from each atom forms the bond. The shared grip on these electrons holds the two atoms together.
|
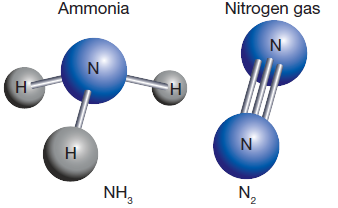
Non-metals only share enough electrons to fill their outer shell or to have eight electrons in it. For example, three additional electrons would fill the outer-shell of nitrogen (electron configuration 2,5). Therefore, a nitrogen atom must pair up three of its electrons with three electrons from other non-metallic atoms. This results in three covalent bonds.
The figure on the right shows that nitrogen can form these three bonds in different ways. |
Covalent bonding usually results in the formation of grouping of atoms known as molecules, such as the two molecules shown above.
|
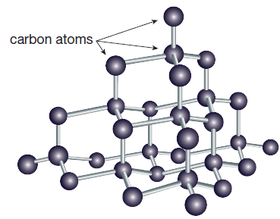
Some elements can use covalent bonding to form extended covalent lattices. The most well known example is carbon that may occurs as lattices of graphite and diamond in pure form along with other less common allotropic forms. The structure of diamond, characterised by the fact that each carbon atom form 4 covalent bonds with 4 neighbouring atoms, is shown on the right. |
Ionic and covalent bonding are further explained and compared in the videoclips below.
|
|
|