writing chemical formulas
The chemical formula of a compound tells you what elements make it up and in what proportions. For example, a molecule of water (H2O) is made of two atoms of the element hydrogen and one atom of the element oxygen. The formula for the ionic compound NaCl (sodium chloride, common salt) indicates that there are equal numbers of ions of the element sodium and the element chlorine.
1. Valency
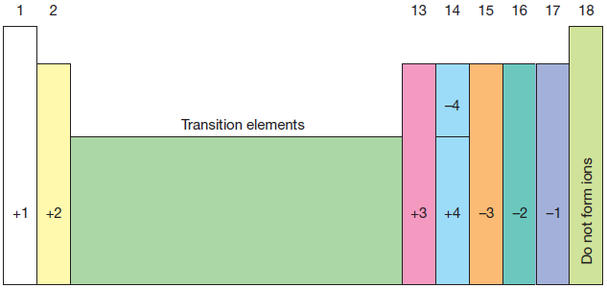
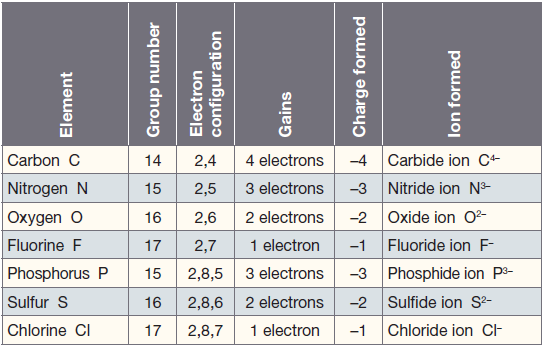
Valency is the combining power of an element. Elements in a group of the periodic table have the same valency. The valency of an element is related to how many electrons are in the outer shell. The table below indicate the valency of any element of groups 1,2, and 13 to 17. The valency of noble gases is 0 as they do not combine with other elements.
Example 1: the valency of elements belonging to group 13 and 15 is 3. Elements of group 2 and 16 have valency of 2.
Example 2: In carbon dioxide (a covalent molecule), carbon has a valency of 4 (group 14) and oxygen has a valency of 2 (group 16).
Example 3: In sodium chloride (an ionic compound), both the sodium ion (group 1) and the chloride ion (group 17) have a valency of 1.
Example 1: the valency of elements belonging to group 13 and 15 is 3. Elements of group 2 and 16 have valency of 2.
Example 2: In carbon dioxide (a covalent molecule), carbon has a valency of 4 (group 14) and oxygen has a valency of 2 (group 16).
Example 3: In sodium chloride (an ionic compound), both the sodium ion (group 1) and the chloride ion (group 17) have a valency of 1.
Note: the valency of carbon can be +4 or -4, depending of the element it combines with.
2. Chemical formula of compounds
|
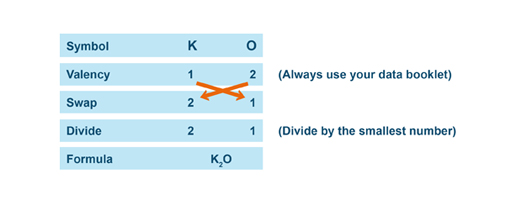
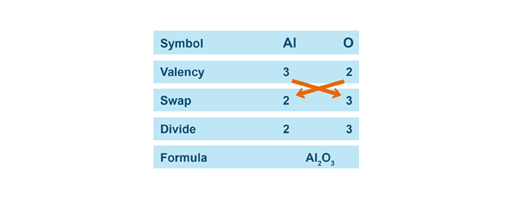
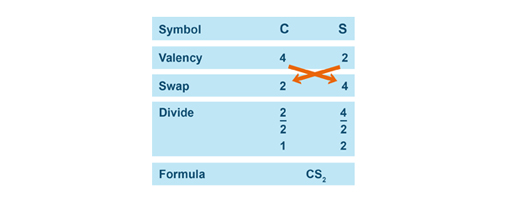
Once the valency of the constitutive elements of the compound are determined, apply the SVSF system to name the compound.
|
S.V.S.F. stands for Symbol, Valency, Swap, and Formula. |
Below are three examples of how to write the chemical formula of each compounds using the SVSF system.