Metallic BONDING
|
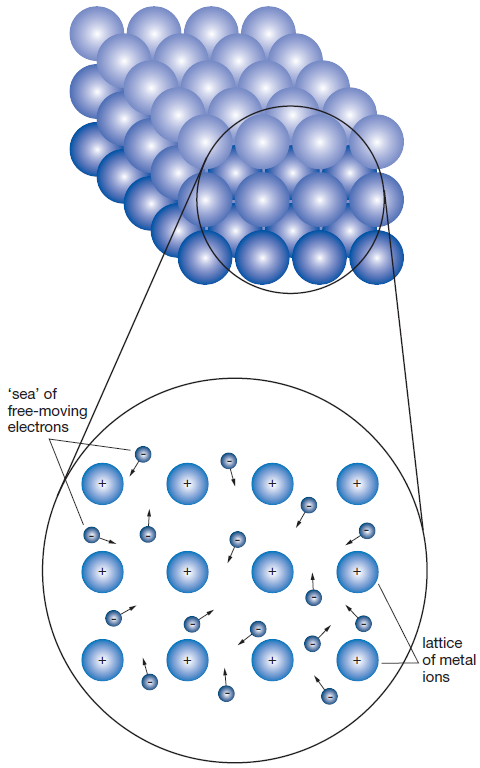
Metal atoms have a weak hold on their outer-shell electrons. This gives the outer-shell electrons the freedom to move throughout the metal without being bound to any one atom. Each metal atom becomes a positively charged ion. Opposite charges attract and this electrostatic force provides three-dimensional bonding between the positive ions and the ‘sea’ of loose electrons surrounding them. This bonding holds the metal together and is known as metallic bonding. A representation of the regular three-dimensional arrangement of cations surrounded by a 'sea' of free electrons is shown on the right.
Metallic bonding explains the main physical properties characteristic of metals:
|
The following video clip explains the nature of metallic bonding and how it accounts for the physical properties of metals.