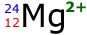What are ions?
When an atom gains or loses one or more electrons while the number of protons stays the same (so that the nature of the element does not change), it becomes an ion. The gain or loss of electrons resulting from a chemical reaction always involves valence electrons. To put it simply,
Ions are atoms with a different number of protons and electrons.
When an atom loses electrons (in other words, when it loses negatively charged particles), it becomes a positively charged ion, or cation (tip: think of the '+' shape of the 't' in the word 'cation' to remember its charge).
When an atom gains electrons, it becomes a negatively charged ion, or anion. Anions always have a larger atomic radius than the atom they originate from.
Ions are atoms with a different number of protons and electrons.
When an atom loses electrons (in other words, when it loses negatively charged particles), it becomes a positively charged ion, or cation (tip: think of the '+' shape of the 't' in the word 'cation' to remember its charge).
When an atom gains electrons, it becomes a negatively charged ion, or anion. Anions always have a larger atomic radius than the atom they originate from.
The video clip below explains how ions are formed from neutral atoms by adding/removing electrons to/from the valence shell.
AZE nomenclature for ions
The AZE system can be extended to cover ions simply by adding the charge onto the element symbol. It is important to remember that a positive ion is the result of a neutral atom having LOST electrons.
For example, consider the ion below:
For example, consider the ion below:
The atomic number is 12 therefore in a neutral atom there would be 12 protons and 12 electrons. However, the charge is 2+ therefore the atom has LOST 2 electrons. The remaining electrons is then 10 electrons.
The same logic applies to negative ions, remembering that a negative ion is the result of a neutral atom having GAINED electrons. For example, when a chlorine atom gains an extra electron, it becomes a chloride ion.
The same logic applies to negative ions, remembering that a negative ion is the result of a neutral atom having GAINED electrons. For example, when a chlorine atom gains an extra electron, it becomes a chloride ion.