Naming compounds
Naming ionic and covalent compounds relies on rules. The rules given below are adapted from this website.
ionic compounds
The simplest ionic compounds are made of only two elements: a metal cation (positively charged) and a nonmetal anion (negatively charged). The following rules apply for naming two-element ionic compounds.
Compounds made of polyatomic ions have more rules:
1. Understand what a polyatomic compound is. These compounds are built off of groups of atoms that are bonded together, and the entire group has a positive or negative charge. You can do three basic things to polyatomic compounds:
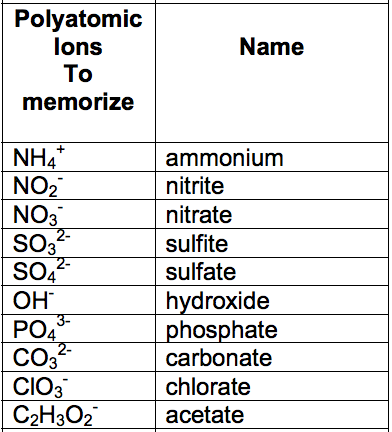
2. Memorize the most common ion groups. These are the basis for forming most polyatomic compounds. A list in order of increasing negative charge is given in the table below.
- Identify the two elements. Use the Periodic Table to find out what groups the elements in the compound belong to.
- Build the name. - The first part of the name is the name of the metal element, unchanged. - The second part is the name of the nonmetal element, modified with “-ide”. Example: Al2O3. Al = aluminium; O = oxygen. So the name would be aluminium oxide.
- Recognize transition metals. Metals groups 3 to 12 of the Periodic Table are known as transition metals. Their charge is written with a Roman numeral when writing out the compound name. This is because they can have more than one charge and make more than one compound. Example: FeCl2 and FeCl3. Fe = iron; Cl = chloride. The names would be iron(II) chloride and iron(III) chloride.
Compounds made of polyatomic ions have more rules:
1. Understand what a polyatomic compound is. These compounds are built off of groups of atoms that are bonded together, and the entire group has a positive or negative charge. You can do three basic things to polyatomic compounds:
- Add a hydrogen to the beginning of the compound. The word “hydrogen” is added to the beginning of the compound name. This reduces the negative charge by one. For example, “carbonate” CO32- becomes “hydrogen carbonate” HCO3-.
- Remove an oxygen from the compound. The charge stays the same and the ending of the compound changes from “-ate” to “–ite”. For example: NO3 to NO2goes from “nitrate” to “nitrite.”
- Replace the central atom with another from the same periodic group. For example, sulfate SO42- can become Selenate SeO42-.
2. Memorize the most common ion groups. These are the basis for forming most polyatomic compounds. A list in order of increasing negative charge is given in the table below.
3. Build compound names based off the list. Associate whatever element(s) is attached to the group ion and name accordingly. If the element comes in front of the ion group, the name of the element is simply added to the beginning of the compound name.
- Example: KMnO4. You should recognize the MnO4- ion as permanganate. K is potassium (as the ion K+), so the compound is named potassium permanganate.
- Another example: NaOH. You should recognize the OH- ion as hydroxide. Na is sodium (Na+), so the compound is named sodium hydroxide.
- One last example: Cu(NO3)2. NO3- is the nitrate ion. Cu is copper (as the Cu2+ ion), so the compound is named copper(II) nitrate, where the roman numeral II indicates the charge of the copper ion.
covalent compounds
Covalent compounds can either be molecular compounds or elements.
Molecular compounds contain at least two different types of atom. We use Greek prefixes to indicate the number of each type of atom in the molecule: mon- for one, di- for two atoms of the same type, tri- for three atoms, tetra- for four atoms, penta- for five atoms, and so on.
Examples:
Some elements exist as molecules made of the same type of atoms. The molecules are sometimes named using the rules for molecular compounds.
Examples:
Some molecules are made of the same type of atoms but are not elements. For example the ozone molecule is made of three oxygen atoms and its formula is O3.
Molecular compounds contain at least two different types of atom. We use Greek prefixes to indicate the number of each type of atom in the molecule: mon- for one, di- for two atoms of the same type, tri- for three atoms, tetra- for four atoms, penta- for five atoms, and so on.
Examples:
- CO: carbon monoxide
- CO2: carbon dioxide
- P2O5: diphosphorus pentoxide
- N2O3: dinitrogen trioxide
- CCl4: carbon tetrachloride
Some elements exist as molecules made of the same type of atoms. The molecules are sometimes named using the rules for molecular compounds.
Examples:
- oxygen O2 (oxygen is the name of the element, dioxygen is the name of the molecule)
- chlorine Cl2 (chlorine: element, dichlorine: molecule)
- hydrogen H2 (hydrogen: element, dihydrogen: molecule)
- nitrogen N2 (nitrogen: element, dinitrogen: molecule)
Some molecules are made of the same type of atoms but are not elements. For example the ozone molecule is made of three oxygen atoms and its formula is O3.