ionic bonding
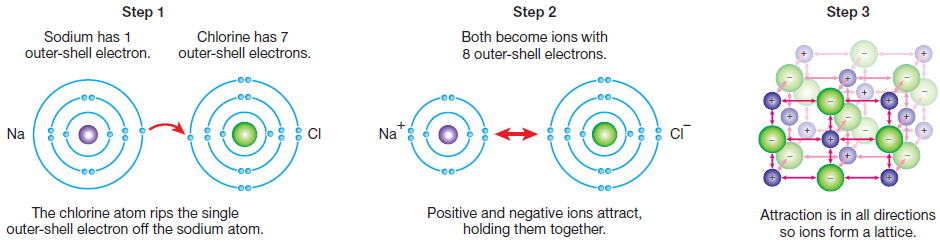
Ionic bonding occurs when metallic elements bond with non-metallic elements. Metal atoms have only a weak hold on their outer-shell electrons. In contrast, non-metallic atoms have a strong hold on their own electrons, and tend to remove outer-shell electrons from any metal atoms nearby. This causes ions to form.
Electrostatic forces pull the positive and negative ions together to form a strong ionic bond. Each ion is surrounded by ions of the opposite charge, building up a three-dimensional structure called a lattice.
Electrostatic forces pull the positive and negative ions together to form a strong ionic bond. Each ion is surrounded by ions of the opposite charge, building up a three-dimensional structure called a lattice.
|
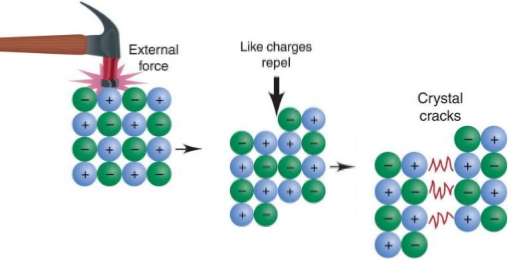
The ionic bonding model explains all the important properties of ionic substances, including how they conduct electricity. When solid, ionic substances don’t conduct electricity because the ions are bonded within their lattice. However, when molten or dissolved in water, these ions separate from one another and are free to move, enabling a closed electrical circuit to be established, as shown in the figure on the right. Another characteristic physical property of ionic compounds is their brittleness. Although they are hard and rigid, they shatter under sufficient external pressure, such as hammering. The figure below explains why this is the case. |