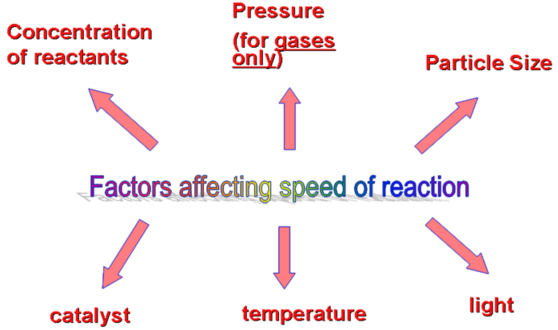
factors affecting rate of reactions
The rate of a chemical reaction depends on a number of factors. The Collision theory account for the way these factors work.
- Temperature: as the temperature increases, the rate of reaction increases.
- Concentration of the reactants: as their concentration increases, the rate of reaction increases.
- Particle size: Increasing the surface area of a solid reactant by increasing the size of particles increases the rate of reaction.
- Catalyst: Catalysts increase the rate of a reaction without being changed themselves by the end of the reaction.
- Pressure of any gaseous reactant: increasing the pressure of a gaseous reactant increases its concentration, therefore increase the rate of reaction.
- Light: The rate of some chemical reactions can be increased by using light with the correct frequency range, usually in the ultraviolet range. Such light break chemical bonds, therefore increasing the rate of reaction.
- Stirring increases reaction rates by moving the reactants' molecules around. For example, when dissolving a sugar (solute) into water (solvent), stirring moves the water molecules around, allowing them to interact with the solid pieces of undissolved sugar and transporting the dissolved solute away into the bulk of the solution.